 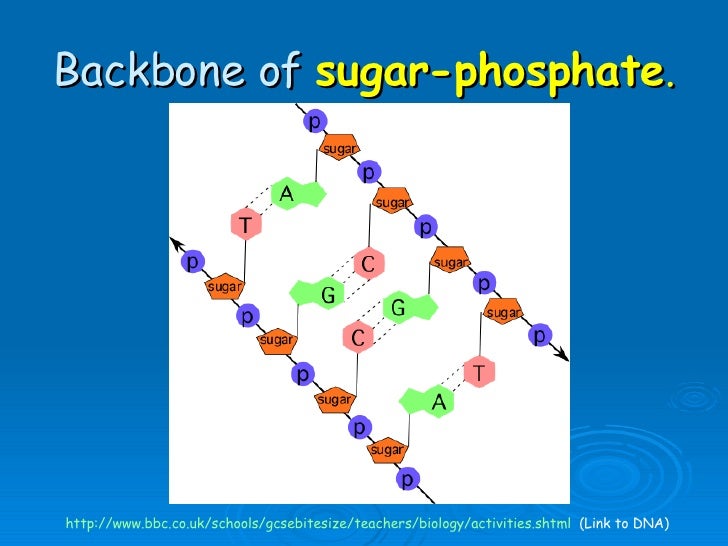
Though the sugar-phosphate backbone is consistent in DNA and RNA, the nucleotide bases vary from one monomer to the next. The chain of sugars connected by phosphodiester linkages is considered as the Nucleic acid backbone. 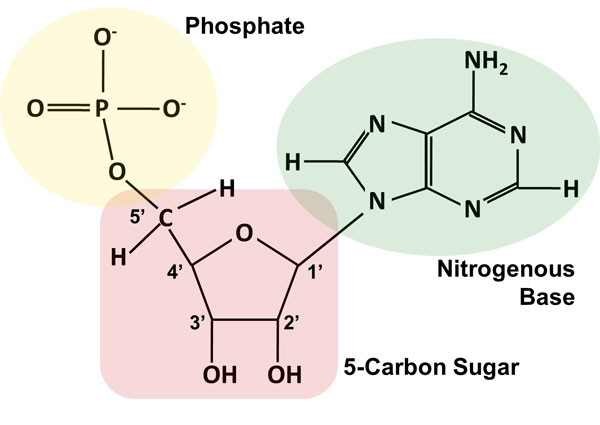
In particular, the 3’ hydroxyl (3 – OH) group of the ribose sugar of one nucleotide forms an ester bond with the phosphate, this phosphate group is also bonded to the 5’ OH group of the adjacent ribose sugar of the neighboring nucleotide. The pentose sugars in nucleic acids are bound to each other by phosphodiester linkages.įigure: Structure of Ribose and Deoxyribose sugar found in RNA and DNA respectively Its prefix deoxy demonstrates that the 2’ carbon atom of the deoxyribose sugar does not have the oxygen atom that is present with the 2’ carbon molecule of ribose sugar (the sugar in ribonucleic acid, or RNA), as displayed in the figure below. The pentose sugar deoxyribose is present in DNA structure (deoxyribonucleic acid). Simple stem-loop structure is observed when a nucleic acid has complementary sequences within the molecule and forms intra-molecular base pairing to form double-helical structures from a single nucleic acid molecule.The replication process is referred to as a semiconservative for DNA.Erwin Chargaff proposed that the proportions of guanine to cytosine and of adenine to thymine were almost something similar in all species taken into consideration.The nucleotide base pairing outcomes in arranging the DNA into a two-strand helical structure.None of the ends is linked to another nucleotide. The opposite terminal or end has a 3 – Hydroxyl group. A DNA strand also has terminals or ends similar to a polypeptide (carboxy and amino terminals). One end or terminal of the DNA strand has a free 5′ – Hydroxyl (or a 5′- hydroxyl group connected to a phosphate group).The N-1 of a pyrimidine or N-9 of a purine is connected to C-1 of sugar.The nucleotide bases are derived from purine guanine (G) and adenine (A), while the other two from pyrimidine uracil (U, RNA just) or thymine (T, DNA just) and cytosine (C).Though the sugar-phosphate backbone is consistent in DNA and RNA, the nucleotide bases vary from one monomer to the next.The chain of sugars connected by phosphodiester linkages is considered as the Nucleic acid backbone.Structural analysis of single stranded DNA.Genetic information carrying capacity of DNA. 
The proposed models are consistent with the current body of experimental data. Owing to this, rotation of one end of the broken sugar-phosphate backbone about the other becomes impossible hence the CPT inhibitory effect on top 1. In the presence of top 1, after CPT conversion to the carboxylate form and DNA nicking, the C17 hydroxyl makes a branching hydrogen bond with N1 and N3 of guanine while the C20 carboxyl makes two hydrogen bonds with the NH of Tyr723 and N(delta2)H(2) of Asp722. The resulting CPT-DNA complex is alkali-labile. As a result, CPT binds to both ends of the broken DNA. In the absence of top 1, the C17 hydroxyl of CPT is involved in ester exchange (nicking of the DNA sugar-phosphate backbone followed by covalent joining of free phosphate to C17) whereas its C20 carboxyl forms two hydrogen bonds with the same guanine nucleotide at the opposite end of the broken DNA backbone. At the second step, CPT is converted to the carboxylate form. 5 (2002)) through hydrogen bonding of its C16a carbonyl with the guanine 2-amino group. At the first step, the CPT lactone form binds to DNA (Streltsov et al., Mol. The proposed models are based on a two-step mechanism of TPT (CPT) dimer interaction with two spatially close DNA duplexes. On the other hand, TPT has been recently shown to bind to DNA. The antitumor activity of camptothecin (CPT) and its derivatives, including water-soluble topotecan (TPT), is determined by their ability to inhibit human DNA topoisomerase I (top 1). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
